Washington — The Supreme Courtroom is about to convene Tuesday to listen to arguments in a case involving a generally used abortion tablet and up to date actions by the Meals and Drug Administration to make the treatment simpler to acquire.
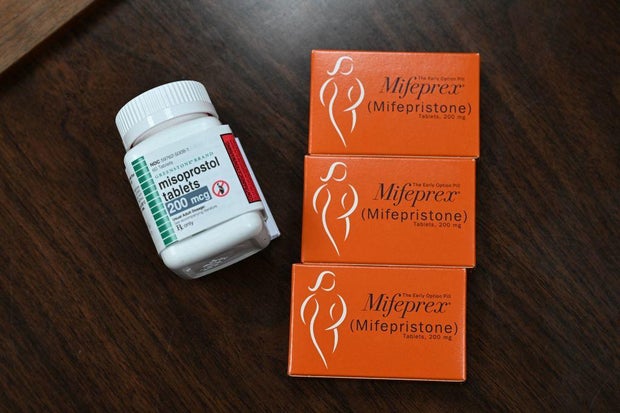
On the middle of the authorized battle is the tablet mifepristone, which is taken together with one other drug to terminate an early being pregnant. Permitted by the FDA in 2000, greater than 5 million sufferers have taken mifepristone, in response to the company, and research cited in court docket filings have proven it’s protected and efficient.
In recent times, the FDA has taken a sequence of steps to make mifepristone extra accessible, together with permitting it to be taken as much as 10 weeks into being pregnant and delivered by means of the mail with out an in-person physician’s go to. These actions, taken in 2016 and 2021, have come underneath authorized scrutiny after a gaggle of anti-abortion rights medical doctors and medical associations claimed the FDA violated the legislation when it relaxed the principles.
The Supreme Courtroom is about to evaluate a resolution from a federal appeals court docket that discovered the company’s actions have been illegal. A ruling unwinding these adjustments would threaten to curtail entry to mifepristone nationwide, even in states with legal guidelines defending abortion entry.
Entry to mifepristone has remained unchanged whereas authorized proceedings within the case have continued, for the reason that excessive court docket issued an order final April preserving its availability. That aid will stay in place till the Supreme Courtroom arms down its resolution, anticipated by the top of June.
ROBYN BECK/AFP by way of Getty Pictures
Arguments within the case are happening lower than two years after the Supreme Courtroom dominated in June 2022 to unwind the constitutional proper to abortion and return the difficulty to the states. And the dispute is just not the one one involving abortion that the justices will take into account inside the subsequent month — a second case includes whether or not federal legislation requires emergency room medical doctors in states that ban abortion to carry out the process on pregnant sufferers whose lives are in danger.
The court docket’s consideration additionally comes on the heels of latest findings that treatment abortions within the U.S. have risen for the reason that Supreme Courtroom overturned Roe v. Wade.
A examine printed Monday within the medical journal JAMA discovered that the variety of self-managed abortions obtained utilizing tablets grew within the six months after the excessive court docket reversed Roe. Analysis from the Guttmacher Institute, a corporation that helps abortion rights, printed final week confirmed that treatment abortions accounted for 63% of all abortions that came about inside the U.S. well being care system in 2023, up from 53% in 2020.
The dispute over mifepristone
The problem to the FDA’s efforts surrounding mifepristone was filed in November 2022 — greater than twenty years after the drug was made out there within the U.S. — by a gaggle of medical associations that oppose abortion rights. Introduced in federal district court docket in Texas, the teams, led by the Alliance for Hippocratic Medication, challenged the FDA’s preliminary 2000 approval and its more moderen adjustments in 2016 and 2021.
As a part of these actions, the FDA allowed mifepristone to be taken as much as 10 weeks right into a being pregnant, as an alternative of seven weeks, diminished the variety of in-person visits required from three to 1, allowed extra well being care suppliers to prescribe the drug and lifted a requirement that or not it’s prescribed in-person.
The organizations, represented by the conservative authorized group Alliance Defending Freedom, claimed the FDA didn’t have the authority to approve mifepristone on the market and didn’t adequately take into account the drug’s security and effectiveness.
The federal decide overseeing the case, U.S. District Choose Matthew Kacsmaryk, agreed that the FDA’s 2000 approval and subsequent actions have been probably illegal. He blocked the FDA’s preliminary motion permitting the drug to be bought within the U.S.
However Kacsmaryk put his ruling on maintain for every week, and a federal appeals court docket and the Supreme Courtroom intervened. The excessive court docket finally maintained entry to mifepristone whereas authorized proceedings continued.
Months later, the U.S. Courtroom of Appeals for the fifth Circuit upheld the FDA’s 2000 approval of the abortion tablet, however mentioned the company violated the legislation with its more moderen adjustments. The appeals court docket’s resolution, although, is preempted by the Supreme Courtroom’s earlier April 2023 order defending entry.
The Justice Division and Danco Laboratories — the maker of Mifeprex, the brand-name model of mifepristone — requested the Supreme Courtroom to evaluate the fifth Circuit’s ruling, and it agreed to take action in December.
The arguments within the case
Celal Gunes/Anadolu by way of Getty Pictures
In asking the justices to reverse the appeals court docket’s resolution, the Biden administration has argued that the medical associations and their doctor members have failed to indicate that they could be injured by the FDA’s actions, and that these alleged accidents might be traced to the FDA’s easing of the principles for mifepristone.
The medical doctors difficult the adjustments don’t prescribe the drug and have not recognized a single case the place a member has been pressured to finish an abortion for a girl who reveals up at an emergency room with an ongoing being pregnant, Solicitor Common Elizabeth Prelogar instructed the court docket in filings.
However legal professionals for the medical teams, represented by the Alliance Defending Freedom, argued that their members object not solely to abortion, but additionally to “complicity within the course of.”
“FDA has spent a long time directing ladies harmed by abortion medication to emergency rooms. Lots of them have sought therapy from respondent medical doctors,” the legal professionals wrote. “Now that FDA known as to account for the hurt brought on, the company can’t insist that the very therapy possibility it directed is in some way speculative.”
If the Supreme Courtroom agrees with the Justice Division that the medical doctors don’t have the correct foundation to sue in federal court docket, it might order the case dismissed with out deciding whether or not the FDA acted inside the bounds of the legislation when it modified the principles for mifepristone’s use.
But when the justices attain the authorized points raised within the case, the Justice Division and Danco have urged the court docket to search out that the FDA’s 2016 and 2021 actions have been lawful.
The company relied on a “voluminous physique of medical proof” on mifepristone’s use over a long time when it decided that the 2016 adjustments could be protected, Prelogar wrote. In any occasion, the district court docket was fallacious to second-guess the determinations that Congress empowered the FDA to make, she mentioned.
“To the federal government’s data, this case marks the primary time any court docket has restricted entry to an FDA-approved drug by second-guessing FDA’s skilled judgment in regards to the circumstances required to guarantee that drug’s protected use,” Prelogar wrote.
Pharmaceutical firms and former heads of the FDA have warned the court docket {that a} resolution upholding the fifth Circuit threatens to undermine the company’s drug-approval course of and will result in persistent authorized challenges of its approval choices.
The decrease court docket’s method, if left intact, “would permit courts to substitute their lay evaluation for FDA’s scientific experience and to overturn the company’s approval and circumstances of use for medication — even after they’ve been in the marketplace for many years,” a gaggle of former commissioners and performing commissioners instructed the court docket in a quick.
“The ensuing uncertainty would threaten the incentives for drug firms to undertake the time-consuming and expensive funding required to develop new medication and finally hinder sufferers’ entry to essential treatments that stop struggling and save lives,” they mentioned.
A slew of pharmaceutical firms and executives individually careworn the significance of drug firms having the ability to depend on the courts to respect the FDA’s scientific judgements.
“If a court docket can overturn these judgments a few years later by means of a course of devoid of scientific rigor, the ensuing uncertainty will create insupportable dangers and undermine the incentives for funding whatever the drug at challenge,” they mentioned in a transient. “This, in flip, will finally harm sufferers.”
However legal professionals for the medical associations and their members that oppose abortion rights argued that the FDA failed to offer a “passable rationalization” for its resolution to carry the in-person shelling out requirement and known as the research the company relied on “deeply problematic.”
Withdrawing the in-person go to requirement in 2021 eradicated the chance for well being care employees to display screen for ectopic pregnancies and different circumstances, the associations argued. In 2016, the FDA eliminated “interrelated safeguards with out research” that examined the adjustments as a complete, they continued.
The group People United for Life, which is backing the Alliance for Hippocratic Medication, claimed that the FDA has promoted entry to abortion tablets with out medical supervision, which have elevated well being and security dangers to ladies and interfered with their care.